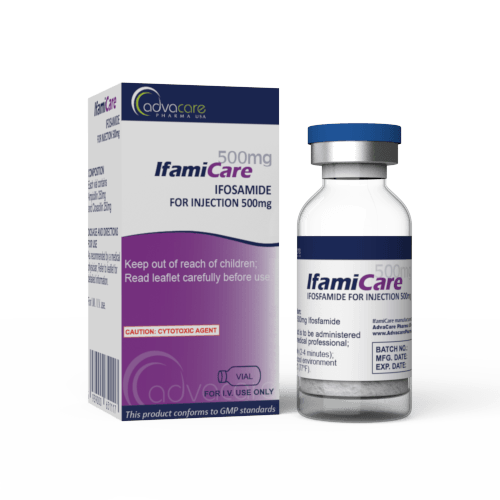
Ifosfamide for Injection
Dosage
Packaging
What is Ifosfamide?
Active Ingredients: Ifosfamide
Ifosfamide for Injection is an anticancer drug used for chemotherapy. When combined with certain antineoplastic drugs, it is indicated for third-line chemotherapy of germ cell testicular cancer. This drug can be used alone to treat various other cancers, including ovarian cancer and osteosarcoma.
The active ingredient, Ifosfamide, is a nitrogen mustard alkylating agent used in the treatment of cancer. Alkylating agents work by slowing or stopping the growth of cancer cells in the body.
This medication is produced and exported by AdvaCare Pharma. Ifosfamide for Injection is manufactured in our facilities in China, India, and the USA. Our factories comply with WHO guidelines and standards, and we regularly inspect these facilities to ensure they meet these high standards.
Why choose us as your Ifosfamide manufacturer?
AdvaCare Pharma manufactures Ifosfamide Injection, one of 40+ oncology treatments, according to rigorous GMP protocols at our state-of-the-art production facility. We are committed to producing quality-assured, cost-effective cancer treatments that meet the highest industry standards and are readily available to cancer patients worldwide. As an established Ifosfamide manufacturer, we supply pharmaceutical distributors, hospitals and government institutions with reliable cancer drugs.
Uses
What is Ifosfamide used for?
It is used to treat cancer. It is typically used in combination with other medicines as a third-line therapy for germ-cell testicular cancer. It can also be used to treat many other types of cancer, such as:
- breast cancer
- lymphoma
- cervical cancer
- ovarian cancer
- osteogenic sarcoma
- soft tissue sarcoma
- bone cancer
- bladder cancer
- small cell lung cancer
How should Ifosfamide for Injection be used?
This medication is manufactured as a powder and stored in a vial. The powder is to be prepared into a solution, and after reconstitution, it should be administered intravenously.
Caution should be used when preparing and administering the solution, and goggles, gloves, and protective gowns are recommended.
What dosage should be given?
The usual dose for adults is 1g, given as a single dose.
The exact dosage is dependent upon the individual patient and treatment plan. Refer to a doctor or pharmacist for guidelines on dosage. Do not exceed what they advise.
Side Effects
As with all pharmaceuticals, some unwanted effects can occur from the use of Ifosfamide for Injection.
Common side effects include, but may not be limited to:
- fatigue
- alopecia
- nausea and vomiting
- diarrhea
- anemia
Seek medical attention if the following develop:
- dermatitis
- stomatitis
- cardiotoxicity
For a comprehensive list of all possible effects, consult a doctor.
If any symptoms persist or worsen, or you notice any other symptoms, please call your doctor immediately.
Precautions
Do NOT use Ifosfamide for Injection if:
- You suffer from any cardiac disease.
- You suffer from peripheral neuropathy.
- You suffer from anorexia.
Consult with your doctor about any medications you are taking before your treatment with Ifosfamide.
There is little evidence with regards to the use of Ifosfamide during pregnancy or breastfeeding, so consult a doctor or healthcare professional before treatment with Ifosfamide for Injection.
During treatment, alcohol should be avoided, as it may worsen nausea and vomiting caused by Ifosfamide. Grapefruit reduces the efficacy of Ifosfamide, so grapefruit products should also be avoided during treatment.