How can we help?
Our Business Model
What is the AdvaCare Pharma business model and what sets it apart within the medical products industry?
We work directly with medical distributors, who are engaged in the importation and/or distribution of pharmaceuticals, medical devices, supplements and veterinary medicines, by supplying quality-assured products adapted to the needs of each specific market.
AdvaCare Pharma has implemented a unique business model that represents a comprehensive methodology and philosophy resulting in highly collaborative business relationships. Our vested supplier-distributor relationship emphasizes a shared commitment in which both parties are equally committed to each other’s success.
By adopting our vested approach, medical distributors become part of an environment that encourages innovation. This leads to enhanced service quality, cost reduction, and the creation of value that was previously untapped, benefiting both parties involved.
How does your unique business model benefit medical distributors seeking a collaborative partnership beyond the traditional buyer-seller dynamic?
- Prioritizes outcomes over transactions
- Concentrates on defining ‘what’ needs to be achieved, rather than 'how'
- Agrees upon well-defined, measurable outcomes
- Implements a pricing model with incentives to optimize business performance
- Adopts an insight-driven approach rather than one based solely on oversight.
What is the process for collaborating with AdvaCare Pharma as a distributor, and what are the requirements for partnership?
- Distributor Company Information: legal status, scope of business, market position and distribution channels
- Product Registration: required documentation, sample requirements and overall registration procedures
- Distribution Terms: payment terms, MOQs, importation procedures, warehousing conditions, distribution methods and sales projections
Although we do have pre-defined criteria that must be met by all new distributors, we do take into consideration the challenges and realities of each market. We realize that every distributor has unique needs and we remain flexible to adapt to local conditions.
Contact us to apply for distributorship of AdvaCare Pharma products.
Products
What types of products does your company specialize in?
- Pharmaceuticals: common generics, specialty generics, OTC, oncology
- Medical Devices: injection instruments, diagnostic detection, medical disposables, skin & wound care, incontinence & urology
- Supplements: general health, condition-specific, vitamins & minerals, herbal formulations
- Veterinary: veterinary generics, custom veterinary formulations, animal nutritional supplements, veterinary vaccines, animal medical devices
You can see all product divisions here.
What makes your product range different from other manufacturers?
- Pharmaceuticals: Comprising 500+ products across 13 dosage forms, our range fulfills the broad requirements of hospitals, clinics, pharmacies and other medical institutions.
- Medical Devices: 5 product divisions, including 500+ class I & II devices with 1M+ specification options, are positioned to meet the everyday needs of hospitals, clinics, pharmacies and labs.
- Supplements: 150+ scientifically-formulated nutritional products manufactured in 9 dosage forms provide a vast product selection to fulfill the consumer demands of every market.
- Veterinary: Encompassing 250+ generic and custom formulations in 13 dosage forms, our livestock and pet treatments empower veterinarians, vet supply retailers and livestock farmers to address animal healthcare challenges.
Our product efficacy, packaging and global brand reputation reflect a higher standard that medical professionals and consumers in more than 65 countries have come to trust.
How to access the technical specifications of a product?
You can access technical specifications and comprehensive product details by visiting the dedicated webpage of a specific product or by contacting us for product-based inquiries.
I cannot find the product specification that is needed for our market.
If a product specification is not available on the product web page or in our catalog, contact us to inquire about specification options.
Can a product be customized to meet the needs of our market?
In many cases, we can customize product specifications according to specific requirements. AdvaCare Pharma utilizes advanced agile manufacturing methods to adjust our production lines to meet the product demands of global markets.
Contact us to inquire about product customization.
How can customers verify if the AdvaCare Pharma products they received are authentic?
The safety and efficacy of our products is our highest priority. We have various measures implemented to verify product authenticity and identify counterfeit products.
Email us directly for product verification or to report a counterfeit product.
How can we obtain assistance with the administration or usage of a product we have purchased?
Our product web pages provide extensive details about each product. However, healthcare-related questions and issues are only to be advised by a medical professional who is qualified and fully familiar with the status of your healthcare situation and/or condition. Contact your healthcare provider for administration and usage questions and advice.
Do you offer discounts or complimentary products for bulk purchases?
AdvaCare Pharma enforces an ethical pricing policy which ensures that all product pricing is calculated according to a strict criteria, including but not limited to API and raw material costs, variable production costs, currency exchange rates and other factors, that prevent any form of price fixing, gouging or manipulation.
We provide extensive support to our medical distributors with beneficial payment terms, free samples, promotional materials, strategic planning, on-the-ground training and numerous other programs to empower their development and bring affordable healthcare to their markets.
Receiving a Quotation
A product inquiry can be submitted by filling the contact form or emailing us directly. However, if you wish to receive a more accurate quotation sooner, submit an online inquiry by adding the products of interest to your inquiry list.
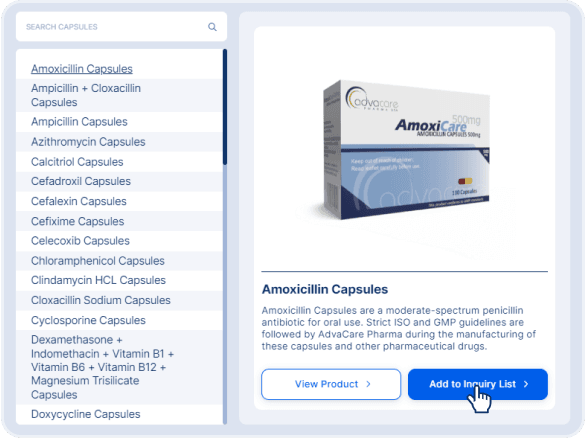
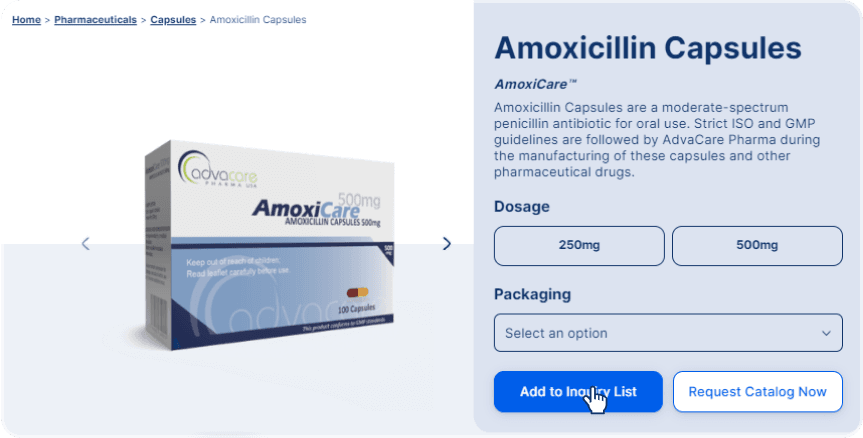
You can access the product navigation pages and directly add products to your inquiry list:
You can also check your inquiry list at any time by clicking on:


Ordering & Shipping
What is the process for placing an order?
We minimize the lead time of every order by following a defined process to identify needed information and ensure accuracy. Our sales specialists walk our distributors through the process efficiently.
After making contact with a sales specialist, simply answer their questions and prepare the requested information. You can contact a sales specialist by filling a short online form.
NOTE: AdvaCare Pharma only sells products in bulk to distributors or retailers. For this reason, we cannot fulfill orders for individual use or for small quantities.
What is the expected delivery time for orders?
As every country has specific requirements for packaging, product specifications, regulatory documentation and other conditions, AdvaCare Pharma manufactures on a per-order basis. On-demand production ensures import and distribution requirements are met and maximizes the shelf-life of every product.
The standard lead time for an order is 30-60 days. However, production and shipping lead times can vary depending on the aforementioned requirements.
What incoterms are available for orders?
The standard incoterm of an order is FOB (Free On Board). Other incoterms, including but not limited to CIF (Cost, Insurance and Freight), depend on various factors and are at the discretion of AdvaCare Pharma.
Is assistance provided with the shipping process?
AdvaCare Pharma provides comprehensive logistical support free-of-charge for our medical distributors. As one of our value-added services, our highly experienced Logistics Department assists with every aspect of getting our products to our distributors in a safe and timely manner.
From where are shipments dispatched?
As a global manufacturer, our production bases span China, India, the United States and other manufacturing locations. Regardless of where our products are shipping from, our medical distributors trust in our control of the supply chain and capability to deliver standardized products on-time and free of damage.
Global Presence
In which countries does AdvaCare Pharma operate?
AdvaCare Pharma currently has distribution of our products in 65+ countries worldwide. Our main markets span countries across Asia, Africa, Latin America & the Caribbean, Middle East, Central Asia (CIS), East Europe and Oceania. We continue a strategic expansion into regions that need our products the most.
See more about our distribution regions.
What if AdvaCare Pharma does not have a presence in our country?
While each country has its own distinct regulations and specific conditions influencing the pharmaceutical and medical sectors, our extensive product range and commitment to regulatory compliance place us in a favorable position to cater to a wide range of markets.
Contact us to inquire about distributing our products in your country.
What makes AdvaCare Pharma the preferred choice for our market?
In a healthcare landscape where cost-effectiveness and product quality are paramount, AdvaCare Pharma offers a distinctive advantage. We excel in meeting the varied demands of most markets by providing competitively priced, high-quality medical products.
Our reputation as a global leader in healthcare is built on trust and recognition from medical professionals and consumers alike. AdvaCare Pharma brands continue to earn widespread acclaim, making us the ideal choice for your country's healthcare needs.
Exclusivity
Is exclusive distribution a possibility in our country?
Yes, we do provide exclusive distribution to carefully chosen medical distributors.
AdvaCare Pharma grants exclusive distribution rights based on several factors which are evaluated according to the distribution country. Exclusive partnerships can provide unique advantages and customized solutions to empower our distributors to succeed in their markets with our products.
Learn more about becoming an AdvaCare Pharma distributor. Or contact us to discuss terms and/or apply for distributorship.
How do we know if there is already an exclusive distributor in our market?
Contact us to inform of your country of distribution and indicate the product range of interest. Our sales specialist will contact you to further discuss distribution options.
If there is an existing exclusive distributor in our country, can our company still distribute AdvaCare Pharma products?
Our extensive product range includes over 4,000 medical products, encompassing pharmaceuticals, medical devices, supplements, and veterinary products. Distribution rights can vary depending on the specific country and the scope of products already in distribution.
Distribution options may still be available in your country. Contact us to inquire further about distribution options in your country.
What's the process for becoming an exclusive distributor?
Our distributor approval process is efficient and transparent. A methodical procedure defines how we grant exclusive rights to qualified distributors. Regardless of the distribution terms with a distributor, our goal is to always protect the interests of the distributor by creating a favorable environment for both market entry and expansion.
Contact us to start the distributor approval process.
Quality & Safety
What quality assurance measures are in place to ensure product reliability?
Our products undergo rigorous quality control procedures and comply with industry regulations and standards, ensuring reliability throughout the supply chain. It is of utmost importance that all of our distributors have complete trust in the AdvaCare Pharma products they distribute.
Learn more about our production and quality standards.
Can documentation of product safety and certifications be provided for compliance needs?
We can provide safety documentation and certifications upon request to meet the specific compliance requirements according to the country of distribution.
Are clinical and non-clinical studies available?
Most products do possess clinical or non-clinical studies. However, the status of available studies, including the applicable phases, vary depending on the product. The compliance requirements for clinical and non-clinical studies also vary according to each country.
We assess the compliance requirements for every country before initiating the product registration process. Contact us for more information about regulatory compliance.
Manufacturing
Where are AdvaCare Pharma products manufactured?
Our products are manufactured in our state-of-the-art facilities located in the US, China, India, Malaysia and Vietnam. We adhere to strict quality control and compliance standards in our production processes.
What is the production capacity of AdvaCare Pharma?
As our product ranges include more than 4,000 medical products spanning pharmaceuticals, medical devices, dietary supplements and veterinary, production capacity varies depending on the facility and production line.
Prior to initiating product registrations or orders starting production, we carefully assess production capacity to ensure timely delivery of all orders.
Does AdvaCare Pharma offer CMO or CDMO services?
Although we do provide some CMO (Contract Manufacturing Organization) services depending on the distributor and market, the majority of our manufacturing capacity is allocated for production of AdvaCare Pharma brands.
We do provide CDMO (Contract Development and Manufacturing Organization) services for distributors and markets that require specific formulations or customized products for increased market adaptation.
Does AdvaCare Pharma offer OEM services?
In the pharmaceutical industry, OEM (Original Equipment Manufacturer) usually refers to the production of machinery and equipment for production. We do not manufacture or supply such products.
However, OEM not specific to the pharmaceutical industry often refers to a contract manufacturer that produces products according to customer requirements. While the majority of our products are only produced under our brands to ensure quality, authenticity and protect the market position of our distributors, specific markets require variations in product specifications or brand names in which case we work with our distributors to adapt our products to their market.
See more about our brands or contact us to learn more about adapting our products for your market.
Product Registration
We want to start product registration in our country, but not sure how to start. What should we do?
- Check online for the official website of the MOH, FDA or other health regulatory agency in your country.
- Locate the medical product registration guidelines.
- If cannot locate this information or the website does not contain this information, contact the MOH, FDA or other health regulatory agency in your country.
- Unless your company has in-house regulatory affairs personnel, we strongly suggest consulting with a third-party registration specialist who knows the local regulations well and can assist to complete the registration process accurately and efficiently.
What is the product registration process when dealing with AdvaCare Pharma?
The first step for a medical distributor is to determine the product(s) needed for distribution in their market. This may seem an obvious initial stage, but extensive market research needs to be conducted to determine sufficient product demand, product positioning and product mix. As significant time and resources will be expended by both AdvaCare Pharma and the distributor throughout the registration process and in preparation of market entry, it is crucial that the distributor carefully chooses the right product(s) to ensure successful distribution.
Once the above is effectively completed by the distributor:
- Contact us to inform the product(s) of interest for distribution. Alternatively, if products are selected on our website, submission of an online inquiry may expedite the process.
- Our sales specialist will verify the registration requirements and process.
- Our sales specialist will work together with a new distributor to formulate an estimated timeline and cost analysis to complete the registration process.
- A distribution agreement will be finalized to clarify the terms and conditions of distribution rights of AdvaCare Pharma products.
- Our sales specialist will coordinate our RA Department and other internal departments to prepare documentation, product samples and other requirements necessary for registration submission. This process may require a period of time but is necessary to avoid future deficiencies that could delay the completion of product registration.
What certifications is AdvaCare Pharma able to provide?
Certifications provided for registration purposes are dependent on specific facilities, production lines, product ranges and/or products. According to the registration requirements of each country, our sales specialist coordinates with our Regulatory Affairs Department to meet the certification requirements ensuring compliance.
Some of the certifications we can provide, dependant on the specific facility, production line, product range and/or product, include but are not limited to:- Pharmaceuticals: GMP, ISO, WHO-COPP, FSC, COA, COO
- Medical Devices: CE, ISO, USFDA 510(k), FSC, DOC
- Supplements: HACCP, ISO, NSF, BRC, Halal, FSC, COA
- Veterinary: GMP, ISO, COPP, FSC, COA, COO
Contact us to learn more about certifications for product registration.
Can AdvaCare Pharma provide the technical documents required for our country?
Yes, we provide technical documentation according to the registration requirements of each country. However, the technical documents provided must reflect the specific facility, production line and/or product being registered. Therefore, it is crucial that we first identify the complete and accurate requirements with the assistance of the distributor as they are on-the-ground and have access to their local MOH, FDA or other health regulatory agency.
We want distributors to know that the initial process of preparing the requirements and compiling the necessary technical documents for registration submission requires time and patience to avoid future queries, deficiencies and rejections. First-time registrations usually require additional time for preparations, but subsequent registrations most often proceed more quickly once the process is documented and can be replicated.
We are experts in the product registration process, supported by a robust Regulatory Affairs Department and other technical teams that have vast experience having registered thousands of products in more than 65 countries. Contact us to learn more about technical documents for product registration.
How long does it take for AdvaCare Pharma to prepare the registration documents?
- The complexity of the registration requirements.
- The number of products being registered.
- The diversity of products being registered.
Although many of the documents required for registration are standard documents, some may require special preparation. In addition, every country has specific requirements for how the documents shall be compiled for submission. Even harmonized schemes, such as PIC/S (Pharmaceutical Inspection Convention and Pharmaceutical Inspection Co-operation Scheme), ICH (International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use), WHO Prequalification Program and others, in practice can vary from country to country.
Contact us to learn more about the timeline for product registration.
API
Does AdvaCare Pharma supply APIs (Active Pharmaceutical Ingredients) in bulk?
Generally, we do not supply APIs (Active Pharmaceutical Ingredients) or raw materials. In special cases, we do offer API to our existing medical distributors who have an established distribution network of finished products.
Our primary product offerings consist of finished pharmaceuticals, medical devices, supplements, veterinary medicines and other healthcare products.
From where does AdvaCare Pharma source APIs and other raw materials?
We source our APIs and raw materials from reputable manufacturers and suppliers located worldwide. Exceedingly stringent quality control measures, including rigorous testing of all sourced materials, are maintained to ensure every API and raw material meets our high standards for safety, purity, and efficacy before they are used in the production process.
Marketing Support
Does AdvaCare Pharma provide marketing support to help promote our business and the distribution of products in our market?
Yes, we provide extensive marketing support free-of-charge to our medical distributors.
AdvaCare Pharma has implemented a unique “vested supplier-distributor relationship”, an established business model and methodology focused on creating a highly collaborative win-win business relationship. Opposed to the conventional approach of focusing on transactions as the basis of the relationship, our core tenet is a strategic relationship based on outcomes. To this end, we support our distributors with marketing materials, strategic support and training for how to effectively market our products to the target audience.
Contact us to learn more about how we provide marketing support for our distributors.
What marketing materials or resources are available to help us introduce AdvaCare Pharma products in our market?
- printed materials
- office supplies
- staff uniforms
- visual merchandising
- promotional gifts
- digital activations
We also furnish tailor-made strategic plans and initiatives, as well as on-the-ground training, for facilitating market entry and expansion. Our highest priority is to empower the success of our distributors in their markets.
Contact us to learn more about the marketing materials and resources we provide.
Can we collaborate on co-branded marketing campaigns or initiatives to target specific markets and industries?
Absolutely, we're open to collaborative marketing initiatives with our medical distributors. While our product brands are globally marketed and distributed, some brand names are adapted to the requirements and needs of specific markets.
Additionally, we customize our marketing materials to include the branding of our distributor (logo, contact information, etc.) for increased brand awareness and channel promotion to more effectively reach the target audience.
Payment
What are the possible payment terms and payment methods of AdvaCare Pharma?
Our payment terms for B2B/bulk orders vary depending on various circumstances, including, but not limited to, the country*, the distributor, the order and/or the products. The acceptable payment terms and methods of payment are indicated in a Distribution Agreement, which is finalized with every distributor prior to distribution, to ensure clarity, transparency and protection for both parties.
Contact us to learn more about payment terms and payment methods.
*Due to international regulations and trade compliance laws, we are unable to accept payments from or engage in transactions with individuals, organizations, or entities residing in countries that are subject to trade sanctions, embargoes, or other regulatory restrictions. We regularly review and update our compliance policies to adhere to these legal requirements.
How to ensure that our payments are being made to a legitimate bank account and not falling victim to scams?
Ensuring payment security is a top priority of our finance department. We have various security measures in place to prevent fraud, including a thorough verification process for distributors to follow before a financial transaction is initiated. Digital authentication tools also ensure a safe and secure transaction process.
Payments should never be made to a new or different unverified bank account. Contact us or your sales specialist in the event of any questions or concerns prior to initiating a payment.
Customer Support
How can I contact the customer support team?
We excel at providing the highest level of customer service in the medical products industry.
- Business Inquiries: We can be reached by email, phone, WhatsApp, online chat or various social media channels. However, the fastest and most efficient way to contact us is through our online contact form. See all forms of contact on our Contact Us webpage.
- Patient/Consumer Inquiries: For any questions related to the use or local availability of our products, contact a local distributor or your healthcare provider.
- Career Inquiries: If interested in exploring career opportunities at AdvaCare Pharma, visit the Contact Us page to fill an online form and submit your CV/resume.
I am in communication with a person who claims to be a sales representative of AdvaCare Pharma. How do I verify this information?
- Double-check the domain of the email address: [salesperson name]@advacarepharma.com
- Send us an email at [email protected] to verify the authenticity of a salesperson or email address.
- Contact us on WhatsApp to verify the authenticity of a salesperson or email address.
I wish to no longer receive AdvaCare Pharma newsletters. How do I unsubscribe?
When you request information or sign-up for an AdvaCare Pharma program or service, you’re given the option of receiving recurring informational/promotional emails from us. We also give you unsubscribe (opt-out) instructions in all of our communications and always honor all opt-out requests.
You may also fill and submit the unsubscribe form to opt-out now.
The information contained within this page does not constitute company policy and is subject to change at any time. For more specific or detailed questions, contact us.
