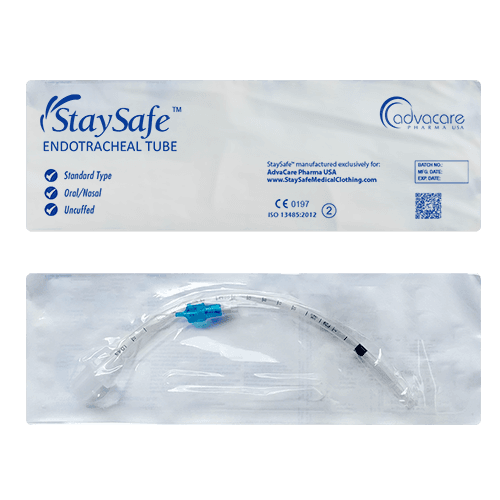
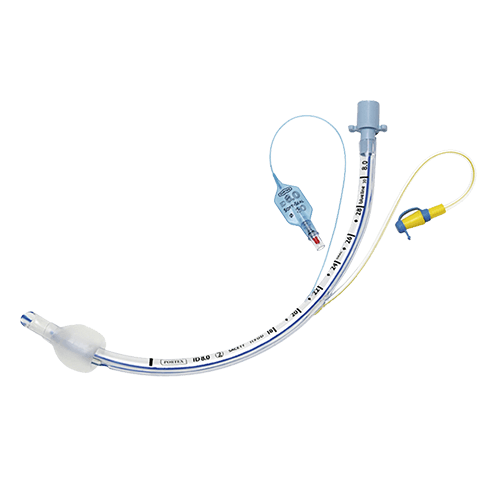
Endotracheal Tube (ET Tube)
Type
Route
Cuff
Inner Diameter
Packaging
What is an Endotracheal Tube (ET Tube)?
An Endotracheal Tube (ET Tube) is a flexible tube that can be inserted into the trachea through the mouth or nose in order to establish an open airway and facilitate mechanical ventilation. This instrument is commonly used during surgeries, emergency situations, or for patients requiring prolonged ventilatory support.
The proper placement of the ET tube is crucial, as it allows for the delivery of oxygen and the removal of carbon dioxide. It is essential to regularly monitor and maintain the tube position for proper ventilation and patient safety.
ET tubes are available in different types, including standard, preformed, and reinforced, and are produced for the oral, nasal, or through either route. It is available in a wide range of diameters, from an inner diameter of 2.5mm to 10mm, to accommodate different patient anatomies. The tube can be produced as either cuffed or uncuffed, with the cuff acting as a seal to prevent air leakage and aspiration.
Endotracheal Tubes (ET Tubes) are manufactured by AdvaCare Pharma in facilities located in China, India, and the USA. These factories strictly adhere to the rigorous regulations required for CE/ISO certification, ensuring the highest quality, safety, and efficiency of our medical products.
Product Specifications
Type
Route
Cuff
Standard
Standard endotracheal tube is commonly used for oral intubation. It is manufactured from clear, flexible plastic.
The tube is available in different sizes to ensure proper fit and accommodate various patient anatomies. Depending on the specific needs of the procedure, the tube can be cuffed or uncuffed, which provides options for controlling airflow and preventing aspiration.

Preformed
Preformed endotracheal tubes are specially designed to match the anatomical contours of the patient's airway. This design is particularly useful in cases where patients have challenging airway anatomy or during procedures that require specific positioning or angulation. The preformed shape of the tube helps to ensure the proper placement and secure airway management.
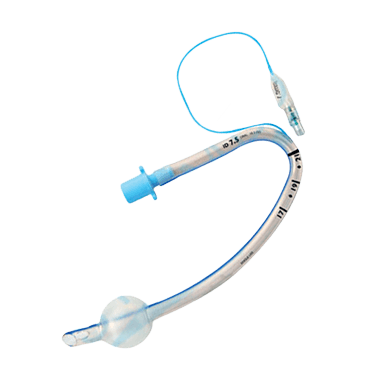
Reinforced
Reinforced endotracheal tube features an added layer of wire within the tube wall, which offers enhanced rigidity and prevents the tube from kinking or compressing. This reinforcement helps to maintain a consistent flow of gases during ventilation.
Reinforced endotracheal tubes are suitable for situations when there is a need for improved stability and durability, such as during long-term intubation or a procedure that requires frequent repositioning of the patient.
Oral/Nasal
Oral/Nasal endotracheal tubes can be inserted through the mouth or nose into the trachea. The versatility of these tubes allows healthcare professionals to choose the most suitable route based on individual patient anatomy and specific clinical requirements.
It is important to note that the intubation technique may differ depending on the route chosen. A patient's airway must be carefully assessed before choosing the appropriate route to ensure effective and safe intubation.
Oral
Oral endotracheal tubes can be inserted through the mouth into the trachea. These tubes are available in different sizes and shapes in order to accommodate many types of patient anatomies and clinical needs.
Depending on the specific requirements of the procedure, oral endotracheal tubes can be either cuffed or uncuffed.
Nasal endotracheal tubes are inserted through the nose into the trachea. They offer an alternative route for airway management and may be used in patients who cannot tolerate oral intubation or when access to the mouth is restricted, such as in certain surgical procedures or cases of facial trauma.
These tubes are available in different sizes and shapes in order to accommodate many types of patient anatomies and clinical needs.
Depending on the specific requirements of the procedure, oral endotracheal tubes can be either cuffed or uncuffed.
Nasal
Nasal endotracheal tubes are inserted through the nose into the trachea. They offer an alternative route for airway management and may be used in patients who cannot tolerate oral intubation or when access to the mouth is restricted, such as in certain surgical procedures or cases of facial trauma.
These tubes are available in different sizes and shapes in order to accommodate many types of patient anatomies and clinical needs.
Depending on the specific requirements of the procedure, oral endotracheal tubes can be either cuffed or uncuffed.
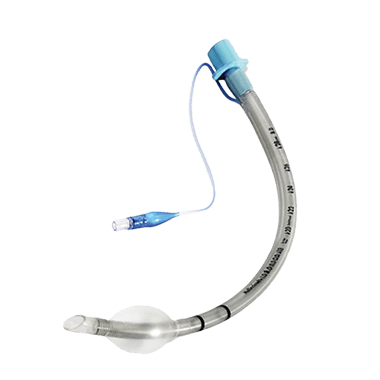
Cuffed
Cuffed ET tube has a small balloon-like structure at the distal end that can be inflated with air to create a seal around the trachea. The cuff is designed to prevent air leakage during mechanical ventilation and to reduce the risk of aspiration of fluids or secretions from the oropharynx or stomach.
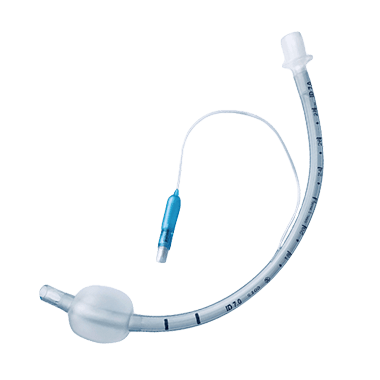
Uncuffed
Uncuffed ET tube does not have an inflatable cuff at the distal end and relies on the natural seal of the trachea to prevent air leakage. It is commonly used in pediatric patients or in situations where a cuffed tube may not be necessary, such as during short-term procedures or in patients with normal lung function.

Why are we a top Endotracheal Tube (ET Tube) manufacturer?
AdvaCare Pharma is an American-owned and operated company specializing in medical supplies for respiratory use. We manufacture Endotracheal Tubes (ET Tubes) and our entire StaySafe™ medical disposables range under strict international guidelines at one of our CE and ISO-certified facilities.
Our distributors have confidence in the quality and reliability of our products, making AdvaCare Pharma a leading supplier in countries where dependable medical supplies and pharmaceuticals are highly sought after. For the past 20 years, we have established a reputation as a trusted supplier of medical products in 65 countries.
Uses
What is an Endotracheal Tube (ET Tube) used for?
An Endotracheal Tube (ET Tube) is used in medical procedures for establishing and maintaining an open airway. This device is commonly utilized during surgeries, emergency medical interventions, or in intensive care settings where patients may require mechanical ventilation.
Through carefully inserting the ET tube into the patient's trachea through either the mouth or nose, healthcare professionals can obtain a secure air passage for the delivery of oxygen and the removal of carbon dioxide. This instrument is indispensable in safeguarding adequate oxygenation and ventilation.
Are there any special storage instructions for Endotracheal Tubes (ET Tube)?
Endotracheal Tubes (ET Tubes) should be stored with utmost care to preserve their integrity and safety. Shield them from direct sunlight and extreme temperature variations, which could compromise their quality.
These tubes should be maintained within their original packaging or placed in suitable containers designed to protect them from potential contamination and physical damage. Adequate storage guarantees that ET Tubes remain sterile and ready for use when needed in medical procedures.
How are Endotracheal Tubes (ET Tubes) used in hospitals? Endotracheal Tubes (ET Tubes) are used in hospitals and clinical settings for various medical applications:
- Surgical Procedures: ET Tubes are commonly used during surgical procedures that require general anesthesia. They supply a patent airway, allowing the administration of anesthetic gases and oxygen to keep the patient safely sedated and ventilated.
- Emergency Situations: In emergency departments, ET Tubes are employed to secure the airway of critically ill or injured patients who may have difficulty breathing independently.
- Intensive Care Units (ICUs): ICUs often utilize ET Tubes for patients requiring prolonged mechanical ventilation. The tubes clear the way for controlled and monitored breathing, especially for patients with severe respiratory conditions.
- Postoperative Care: After surgery, ET Tubes may be retained temporarily to assist with breathing until the patient is fully recovered and can breathe independently.
- Patient Monitoring: In some cases, ET Tubes are used for continuous monitoring of respiratory parameters, making them valuable tools for tracking a patient's progress and adjusting ventilation as needed.
- Diagnostic Procedures: ET Tubes may be utilized during certain diagnostic tests or bronchoscopies to gain access to the lower airways for examination or sample collection.
The accurate insertion and management of ET Tubes in hospitals require highly skilled healthcare professionals, such as anesthesiologists, respiratory therapists, and intensive care nurses, who receive specialized training in airway management and intubation techniques. Securing correct placement and continuous monitoring of the ET Tube are central to patient safety and successful medical procedures.
FAQs
What type of Endotracheal Tube (ET Tube) should be used?
Standard ET tubes provide a basic option for airway management. They are versatile and require manual shaping during intubation. Preformed ET tubes have a predetermined curve similar to the natural anatomy of the airway. These tubes facilitate easier and more accurate placement, which helps reduce the need for extensive adjustments. Reinforced ET tubes have an additional layer of reinforcement embedded within the tube wall, adding strength and rigidity to minimize the risk of kinking or collapsing during complex procedures.
What are the benefits of cuffed or uncuffed ET Tubes?
Cuffed tubes are commonly used in surgeries, critical care settings, and situations requiring positive pressure ventilation. Uncuffed ET tubes lack an inflatable cuff and rely on a precise fit between the tube and the tracheal wall. They are suitable for certain pediatric cases, patients with smaller airways, or when cuff-related complications are a concern.
What inner diameter is needed for the ET Tube?
The inner diameter is highly dependent on the patient's age, size, and airway requirements.
Are your ET Tubes compatible with existing healthcare systems?
Our medical devices are meticulously designed and crafted to seamlessly integrate into prevailing healthcare systems and workflows, ensuring effortless adoption and implementation for healthcare professionals.
Are there options to tailor the configurations of your ET Tube to suit my requirements?
We provide options to customize the configurations of numerous medical devices to meet unique user preferences and clinical demands. Leveraging our agile manufacturing techniques, we can enhance production capability to offer a broader spectrum of specifications, ensuring suitability for diverse market needs.
References
Pharmacology in Noninvasive Ventilation in Do-Not-Intubate Patients and Palliative Medicine
This chapter discusses the use of noninvasive ventilation as a palliative strategy for patients who do not wish to undergo endotracheal intubation. It explores the pharmacological considerations in providing comfort without the invasive aspect of ET tubes in certain patient populations.
Early Experience in Use of Videolaryngoscopy by a Neonatal Pre-Hospital and Retrieval Service
This article presents findings on the use of videolaryngoscopy for neonatal tracheal intubation, comparing its success rate to direct laryngoscopy in a pre-hospital and retrieval setting. It highlights the advancements in technologies for securing airways in neonates, emphasizing the importance of ET tube placement.